Announcements
DSRB-271023: Re-Classification of Studies involving Anonymised Data and/or Human Biological Material as Review Not Required
Effective 01 Nov 2023:
- Studies involving anonymised data/ human biological materials will no longer require review by the NHG Domain Specific Review Board.
- This change in policy is aligned with SingHealth and consistent with US Office for Human Research Protections & Health and Human Services positions of defining studies involving anonymised data/HBM as not meeting the definition of human subject research.
Studies Impacted by Policy Change:
- All new studies involving only anonymised data/HBM do not require DSRB review. If submitted to DSRB, the study will receive a Review Not Required Outcome.
- Previously approved applications using anonymised data/HBM may request to be re-classified as Review Not Required (RNR); simply submit a Study Status Report Form (SRF) for DSRB to conduct the re-determination. In the SRF, the researcher should indicate that the study status is “Terminated” and specify that only anonymised data/HBM are studied, thus pending DSRB review to cease oversight. Please submit such SRF requests by 01 Feb 2024.
Impact on Institutions and Researchers:
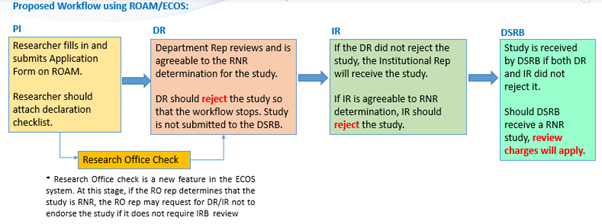
- A declaration form has been co-created with institution research offices to enable self-determination and documentation whether a piece of research involving anonymised data/ HBM can meet the Review Not Required (RNR) criteria.
- Researchers may use the form to evaluate whether their research involving anonymised data/HBM can meet the Review Not Required (RNR) criteria.
- Researchers may subsequently seek endorsement from their department and institutional reps to proceed with their research that meets the RNR criteria and retain the endorsed form as proof of review.
- Institutions may continue to request researchers to submit their research applications on ROAM when there is ambiguity whether the study meets the RNR criteria.
Please click here to download the declaration checklist.
If you have any questions, please contact us at OHRPP@nhg.com.sg.
Diagram 1: